Scientists from the Fritz Haber Institute, Sorbonne University, and Uppsala University have made a discovery that can help to improve our understanding of the behavior of ions in solutions. Their paper, titled “The solvation shell probed by resonant intermolecular Coulombic decay,” has been published in the journal Nature Communications.
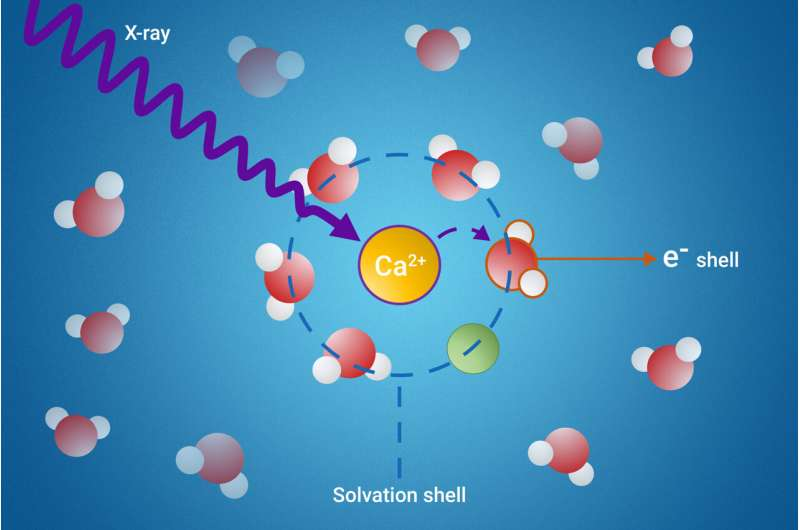
When substances dissolve in a liquid, a solvation shell is formed—a layer of solvent molecules that surround the dissolved particles. The solvent molecules that constitute these solvation shells can acquire wildly different properties from the other, free solvent molecules.
However, studying these solvation shells has always been a challenge due to their complex nature and the difficulty of specifically targeting the solvent molecules that make up the solvation shells but not the many other solvent molecules.
The research team has developed a novel method to probe these elusive solvation shells using a process called resonant intermolecular Coulombic decay (ICD). Their method involves exciting the molecules with X-rays and observing how they interact with their neighbors during the decay process. By doing so, scientists can gain detailed insights into the properties of the solvation shell.
The study revealed that a specific ICD process is a powerful indicator of ion pair formation. The researchers were also able to measure the electron binding energies of water molecules in the first solvation shell. This is a significant achievement, as these measurements were previously unattainable.
Understanding solvation shells is essential for a wide range of scientific fields, including chemistry, biology, materials science, atmospheric science, and electrochemistry. This new method provides a powerful tool for scientists to study these shells in greater detail, which is of relevance for these broad scientific and engineering fields.
More information: Rémi Dupuy et al, The solvation shell probed by resonant intermolecular Coulombic decay, Nature Communications (2024). DOI: 10.1038/s41467-024-51417-3
Journal information: Nature Communications
Provided by Max Planck Society